37% formaldehyde (2.2 mL, 40 mmol),
with stirring until the mixture was completely dark blue solution and then
butylamine (2 mL, 20 mmol) was added with continuous stirring. The mixture of reactants was refluxed for 24 h and
the yellow solution was cooled to room temperature and filtered to remove any insoluble solids. After the amount of
concentrated perchloric acid have been added dropwise, the mixture was filtered and stored at room temperature to
form yellow solids. Yield:
|
67%. The product was purified by recrystallization from acetonitrile : absolute ethanol
in
mole ratio 1 : 1. The physical properties and analytical x-ray data are given in Table 1.
Results and Discussion
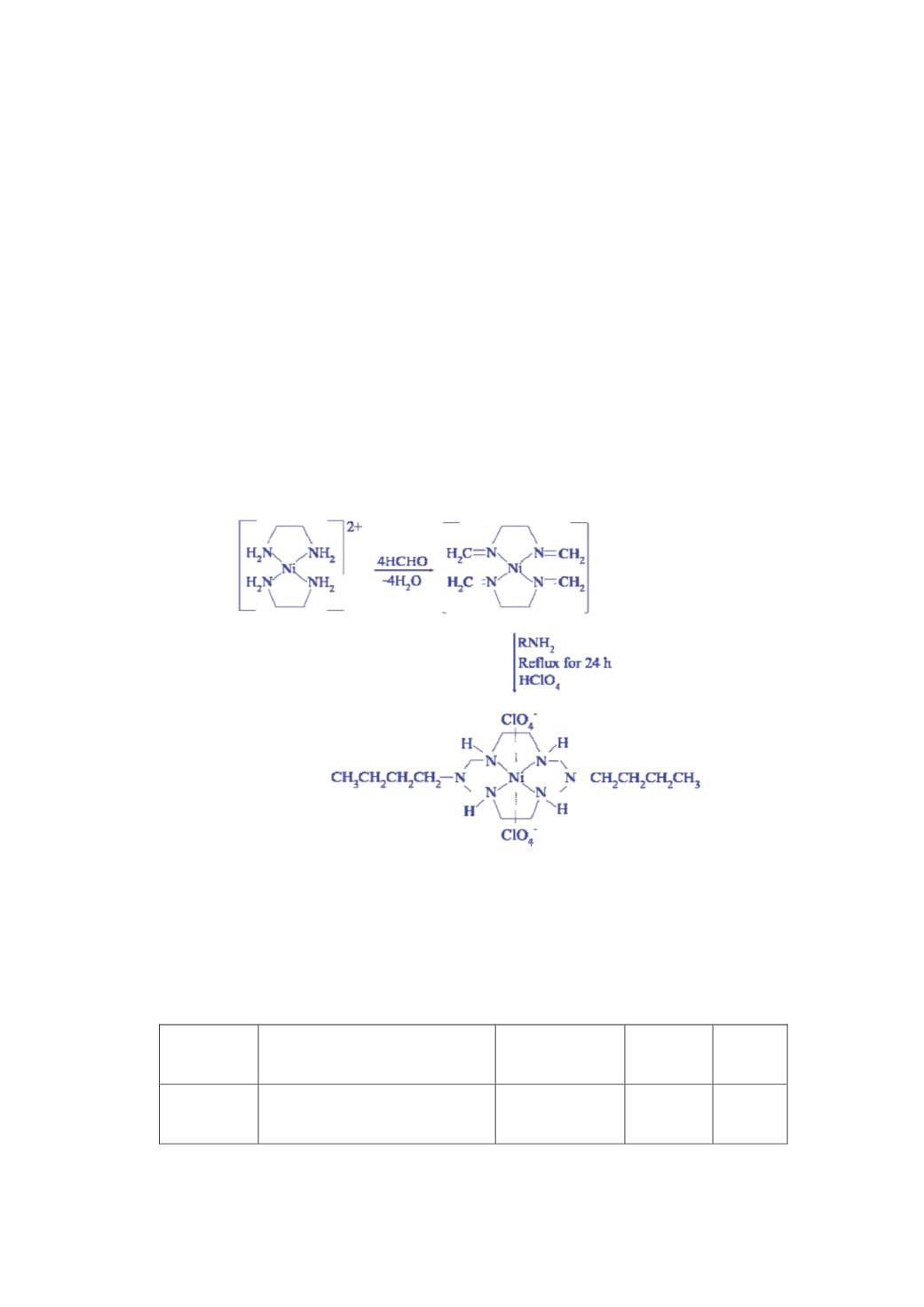
The tetraaza macrocyclic complex has been synthesized in a one – pot reaction involving Nickel chloride,
ethylenediamine, formaldehyde and butylamine as shown in Scheme 1. The synthesized complex is found to be air
stable and insoluble in water, but soluble in some polar solvents, for instance MeCN, DMF and DMSO. The analytical
data of the complexes and some physical properties are summarized in Table 1.
Where L = 1,8-dibutyl-1,3,6,8,10,13-hexaazacyclotetradecane
Scheme 1.
Synthesis of [NiL](ClO
4
)
2
Table 1.
Physical properties and analytical data of nickel(II) complexes containing tetraaza macrocyclic ligands
Complex
Reaction
Empirical
formula
Melting
point (
o
C)
Colour
[NiL](ClO
4
)
2
NiCl
2
•6H
2
O + 2C
2
H
8
N
2
+ 4CH
2
O
+ 2C
4
H
11
N
2
+ HClO
4
C
16
H
38
O
8
N
6
Cl
2
Ni
a
223
yellow
219
การประชุ
มวิ
ชาการระดั
บชาติ
มหาวิ
ทยาลั
ยทั
กษิ
ณ ครั้
งที่
22 ประจำปี
2555


